4-Bromo-(1-oxyl-2,2,5,5-tetramethyl-∆3-pyrroline-3-methyl) Methanethiosulfonate
Share product
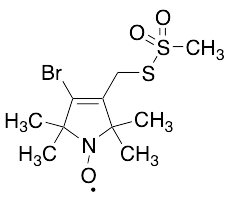
- structure.search.product.name 4-Bromo-(1-oxyl-2,2,5,5-tetramethyl-∆3-pyrroline-3-methyl) Methanethiosulfonate
- Product Code: TRC-B686290
- CAS Number: 215956-55-1
- Brand: TRC
Product Overview
Product Code
TRC-B686290
CAS Number
Product Format
Neat
Molecular Formula
C10 H17 Br N O3 S2
Molecular Weight
343.28
Documentation
Looking for another lot?
To view all certificates of analysis immediately, please login to your account
or
Email download link
{{ errors.first('RequestCoaForm.lotNumber') }}
{{ errors.first('RequestCoaForm.requestEmail') }}
For information about our data processing activities, please visit our Privacy Notice.
Enter your email address and we'll email you the relevant CoA for lots:
{{ coaPopupData.packSize.coaSelectedLotNumbers }}
{{ errors.first('SendDownloadCoaLinkForm.coaEmail') }}
We will be sending the CoA to your email address {{ coaEmailPopupData.userEmail }}
Your request has been sent to our sales team to process.
Find an SDS for your region
{{ errors.first('RequestSdsForm.selectedRegion') }}
{{ errors.first('RequestSdsForm.sdsEmail') }}
For information about our data processing activities, please visit our Privacy Notice.
Your request has been sent to our sales team to process.
Product Information
Chemical Data
Analyte Name
4-Bromo-(1-oxyl-2,2,5,5-tetramethyl-delta-3-pyrroline-3-methyl) Methanethiosulfonate
CAS Number
215956-55-1
Molecular Formula
C10 H17 Br N O3 S2
Molecular Weight
343.28
Accurate Mass
341.9833
SMILES
CC1(C)N([O])C(C)(C)C(=C1Br)CSS(=O)(=O)C
InChI
InChI=1S/C10H17BrNO3S2/c1-9(2)7(6-16-17(5,14)15)8(11)10(3,4)12(9)13/h6H2,1-5H3
IUPAC
3-bromo-1-?^{1}-oxidanyl-2,2,5,5-tetramethyl-4-(methylsulfonylsulfanylmethyl)pyrrole
Product Data
Storage Temperature
-20°C
Shipping Temperature
Room Temperature
Country of Origin
CANADA
Product Format
Neat
Product Description
The Bromo-analogue of MTSL, a highly reactive thiol-specific spin-label. A specific conformational probe of thiol site structure by virtue of its minimal rotational freedom and distance from the covalent disulfide linkage to the macromolecule under study.
References: Berliner, L.J., et al.: Analytical Biochemistry, 119, 450 (1982)
