Nitisinone
Share product
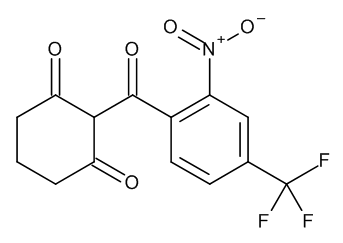
- structure.search.product.name Nitisinone
- Product Code: TRC-N490135
- CAS Number: 104206-65-7
- Brand: TRC
Product Overview
Product Code
TRC-N490135
CAS Number
Product Format
Neat
Molecular Formula
C14 H10 F3 N O5
Molecular Weight
329.23
API Family
NitisinoneProduct Categories
TRC, API Reference Standards & Research Materials, Enzyme inhibitors, Pharmaceutical Toxicology Reference Materials
Purity
>95% (HPLC)
Documentation
Looking for another lot?
To view all certificates of analysis immediately, please login to your account
or
Email download link
{{ errors.first('RequestCoaForm.lotNumber') }}
{{ errors.first('RequestCoaForm.requestEmail') }}
For information about our data processing activities, please visit our Privacy Notice.
Enter your email address and we'll email you the relevant CoA for lots:
{{ coaPopupData.packSize.coaSelectedLotNumbers }}
{{ errors.first('SendDownloadCoaLinkForm.coaEmail') }}
We will be sending the CoA to your email address {{ coaEmailPopupData.userEmail }}
Your request has been sent to our sales team to process.
Find an SDS for your region
{{ errors.first('RequestSdsForm.selectedRegion') }}
{{ errors.first('RequestSdsForm.sdsEmail') }}
For information about our data processing activities, please visit our Privacy Notice.
Your request has been sent to our sales team to process.
Product Information
Chemical Data
Analyte Name
Nitisinone
CAS Number
104206-65-7
Molecular Formula
C14 H10 F3 N O5
Molecular Weight
329.23
Accurate Mass
329.0511
SMILES
[O-][N+](=O)c1cc(ccc1C(=O)C2C(=O)CCCC2=O)C(F)(F)F
InChI
InChI=1S/C14H10F3NO5/c15-14(16,17)7-4-5-8(9(6-7)18(22)23)13(21)12-10(19)2-1-3-11(12)20/h4-6,12H,1-3H2
IUPAC
2-[2-nitro-4-(trifluoromethyl)benzoyl]cyclohexane-1,3-dione
Product Data
Storage Temperature
-20°C
Shipping Temperature
Room Temperature
Country of Origin
CANADA
Product Type
API
Product Format
Neat
API Family
Purity
>95% (HPLC)
Product Description
Nitisinone is a herbicidal triketone that inhibits 4-hydroxyphenylpyruvate dioxygenase (HPPD), an enzyme involved in plastoquinone biosynthesis in plants and in tyrosine catabolism in mammals. It is used in treatment of inherited tyrosinemia type I.
References: Lindstedt, S., et al.: Lancet, 340, 813 (1992), Ellis, M.K., et al.: Toxicol. Appl. Pharmacol., 133, 12 (1995), Lock, E.A., et al.: J. Inherited Metab. Dis., 21, 498 (1998),
