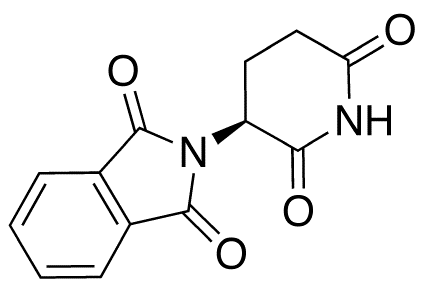
(S)-(-)-Thalidomide
Share product
Product Overview
Product Code
TRC-T338860
CAS Number
Product Format
Neat
Molecular Formula
C13 H10 N2 O4
Molecular Weight
258.23
API Family
ThalidomideProduct Categories
TRC, Chiral Molecules , Approved therapeutics/drug candidates, Angiogenesis, Anti-HIV agents, Impurity Reference Materials
Product Type
ImpurityDocumentation
Looking for another lot?
To view all certificates of analysis immediately, please login to your account
or
Email download link
{{ errors.first('RequestCoaForm.lotNumber') }}
{{ errors.first('RequestCoaForm.requestEmail') }}
For information about our data processing activities, please visit our Privacy Notice.
Enter your email address and we'll email you the relevant CoA for lots:
{{ coaPopupData.packSize.coaSelectedLotNumbers }}
{{ errors.first('SendDownloadCoaLinkForm.coaEmail') }}
We will be sending the CoA to your email address {{ coaEmailPopupData.userEmail }}
Your request has been sent to our sales team to process.
Find an SDS for your region
{{ errors.first('RequestSdsForm.selectedRegion') }}
{{ errors.first('RequestSdsForm.sdsEmail') }}
For information about our data processing activities, please visit our Privacy Notice.
Your request has been sent to our sales team to process.
Product Information
Chemical Data
Analyte Name
(S)-(-)-Thalidomide
CAS Number
841-67-8
Molecular Formula
C13 H10 N2 O4
Molecular Weight
258.23
Accurate Mass
258.064
SMILES
O=C1CC[C@H](N2C(=O)c3ccccc3C2=O)C(=O)N1
InChI
InChI=1S/C13H10N2O4/c16-10-6-5-9(11(17)14-10)15-12(18)7-3-1-2-4-8(7)13(15)19/h1-4,9H,5-6H2,(H,14,16,17)/t9-/m0/s1
IUPAC
2-[(3S)-2,6-dioxopiperidin-3-yl]isoindole-1,3-dione
Product Data
Storage Temperature
-20°C
Shipping Temperature
Room Temperature
Country of Origin
CANADA
Product Format
Neat
API Family
Product Description
Optically active isomer of Thalidomide, which inhibits FGF-induced angiogenesis. Inhibits replication of human immunodeficiency virus type 1. Teratogenic sedative.
References: D??™Amato, r.J., et al.: Proc. Natl. Acad. Sci. USA, 91, 4082 (1994), Makonkawkeyoon, S., et al.: Proc. Natl. Acad. Sci. USA, 90, 5974 (1993), Schumacher, H., et al.: J. Pharmacol. Exp. Therap., 160, 189 (1968)